TOPICS
The sources of the following information, graphics, and designs are all at the end of each lesson.
NUCLEAR CHEMISTRY
What is Nuclear Chemistry?
Nuclear chemistry is the study of how atomic nuclei can interact with one another to form new nuclei. Nuclear reactions change the atom itself. Nuclear reactions, like conventional chemical reactions, are accompanied by energy changes. The energies contained within atom nuclei are hundreds of thousands of times greater than the energies of chemical bonds. To balance nuclear reactions, one must concentrate on the subatomic particles contained within the atom (protons, neutrons, and electrons). Nuclear processes are also classified in their own way.
Atomic Nucleus:
Located at the center of the atomComposed of proton and neutronComprise Atomic MassContains the Nucleons which are proton and neutronNucleons were held by Strong Nuclear Force
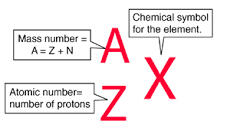
SYMBOL, MASS, AND CHARGE
Proton – p
Mass - 1.673 x 10 1.673 x 10-27kg
Charge – (+) positive
Neutron – N
Mass - 1.675 x 10 1.675 x 10-31kg
Charge - (0) neutral
Isotopes
These are nuclei of the same element with different number of neutron.
• only 250 isotopes are stable
• No stable isotope greater than atoms with atomic number more than 83 (Bismuth). These atoms undergo spontaneous disintegration called decay.
Representation of Element
Radioactivity
The act of emitting radiation spontaneously is referred to as radioactivity. This is accomplished by an atomic nucleus that is unstable for some reason; it "wants" to give up some energy in order to shift to a more stable configuration.
Types of Radioactive Decay
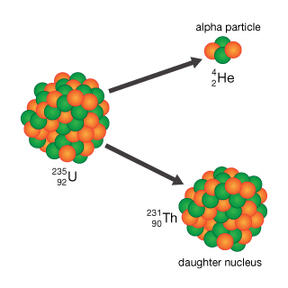
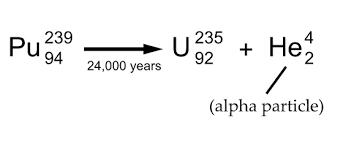
1. Alpha Decay - Disintegration of a nucleus into a nucleus of another element with the emission of alpha particle which is Helium. Refer to the Figure 1.2 for visualization and check the example below.
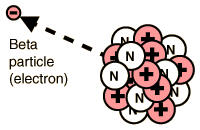
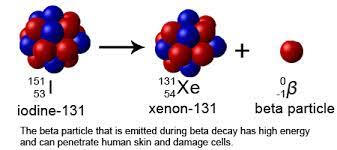
2. Beta Decay - The radioactive transformation of an atomic nucleus accompanying the emission of an electron. It involves unit change of atomic number but none in mass number. Also called: beta transformation or beta process. Refer to Figure 1.3 for visualization and check the example below.
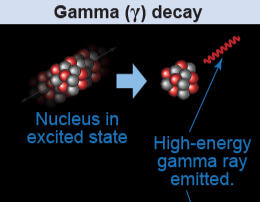
3. Gamma Decay - A type of radioactivity in which some unstable atomic nuclei dissipate excess energy through a spontaneous electromagnetic process Gamma rays (photons, or packets of electromagnetic energy with extremely short wavelengths) are radiated in the most common form of gamma decay, known as gamma emission.
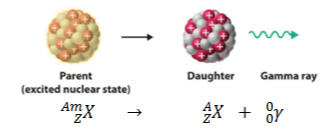
Half Life
In radioactivity, the interval of time required for one-half of a radioactive sample's atomic nuclei to decay (change spontaneously into other nuclear species by emitting particles and energy), or, equivalently, the time interval required for a radioactive material's number of disintegrations per second to decrease by one-half.

https://www.britannica.com/science/gamma-decay
https://ehss.energy.gov/ohre/roadmap/achre/intro92.html
https://www.britannica.com/science/half-life-radioactivity
https://www.britannica.com/science/chemical-bonding
https://courses.lumenlearning.com/boundless-chemistry/chapter/lewis-dot-symbols-and-lewis-structures/
https://chemcenter.ucmerced.edu/sites/chemcenter.ucmerced.edu/files/page/documents/chm002workshop_imf.pdf
https://my.clevelandclinic.org/health/articles/15416-carbohydrates
https://chesquations.wordpress.com/2018/03/12/the-intermolecular-forces-of-attraction/
https://intermolecularforcessite.wordpress.com/2017/01/24/medicine/
https://chesquations.wordpress.com/
CHEMICAL BONDS
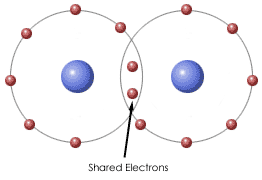
Figure 2.1 Covalent Bond
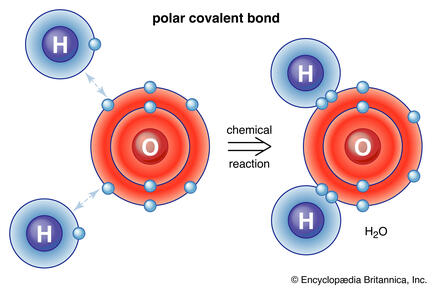
Figure 2.2 Polar Covalent Bond
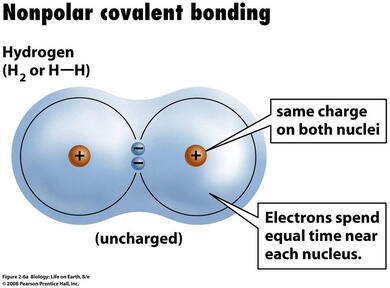
Figure 2.3 Non Polar Covalent Bond
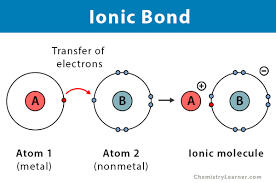
Figure 2.4 Ionic Bond
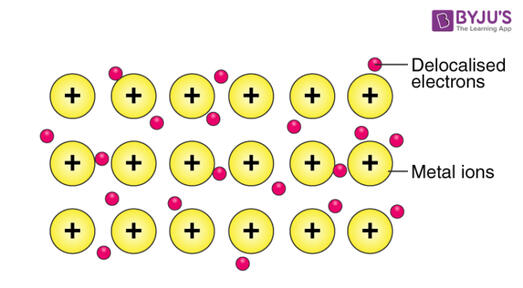
Figure 2.5 Metallic Bond

Figure 2.6 Linear

Figure 2.7 Trigonal Planar

Figure 2.8 Tetrahedral

Figure 2.9 Trigonal Pyramidal

Figure 2.10 Trigonal Bipyramidal

Figure 2.11 Octahedral
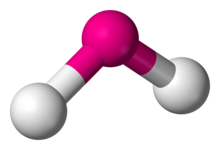
Figure 2.12 Bent or Angular
What is Chemical Bonding?
Any of the interactions that account for the association of atoms into molecules, ions, crystals, and other stable species that make up the familiar substances of everyday life is referred to as chemical bonding. When atoms collide, their nuclei and electrons interact and tend to distribute themselves in space in such a way that the total energy is lower than in any other arrangement. If the total energy of a group of atoms is less than the sum of the energies of the component atoms, the atoms bond together, and the energy lowering is known as the bonding energy.
Electrical attraction between the nuclei and the valence electrons of an atom and which binds atom together. The type of attraction is intramolecular force.
Types of Chemical Bonding
1. Covalent bond - Sharing of electron pair between atoms. This type of bonding occurs between non-metals. When two elements have little difference in electronegativity they usually share an electron. Refer to the Figure 2.1
a. Polar Covalent - Refers to bond in which the bonded atoms have an unequal sharing of electrons. Image on the Figure 2.2
b. Non-Polar Covalent - Refers to bond in which electrons are equally shared by the bonded atoms. Image on the Figure 2.3
2. Ionic bond - Transfer of one or more valence electron from one atom to another. Exist between metal and non-metal. This happens when there is big difference between the electronegativity of two atoms and usually happens between metals and non-metals. Refer to the Figure 2.4
3. Metallic bond - Bonds holding among metal atoms. There are highly mobile valence electrons in the atoms. Refer to the Figure 2.5
Octet Rule
This rule states that an element to be stable there should be 8 electrons in their valence shell. Noble gases who belong to family 8 has 8 electrons in their valence shell/ outermost shell.
Electronegativity
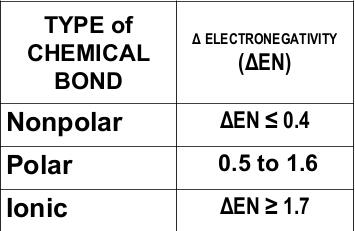
The measure of tendency of an atom to attract electrons towards itself. The absolute value of electronegativity difference (ΔEN) between two atoms gives an idea of the type of chemical bond that can exist between them. We can find the value of electronegativity in Periodic Table of Elements.
VSEPR Theory
Valence-Shell Electron-Pair Repulsion (VSEPR) Theory - This theory assumes that each atom in a molecule will achieve a geometry that minimizes the repulsion between electrons in the valence shell of that atom.
Lewis Dot Structure
Lewis structures (also known as Lewis dot structures or electron dot structures) are diagrams that show the valence electrons of atoms in a molecule. These Lewis symbols and structures aid in the visualization of valence electrons in atoms and molecules, whether they exist as lone pairs or within bonds.
Common Molecular Geometry
1. LINEAR - 2 atoms bonded by central atoms, 0 Lone Pair, 180 degrees. Refer to the Figure 2.6
Type of molecule: AB2
Formula example: BeF2
Lewis Structure
Polarity: Nonpolar
2. TRIGONAL PLANAR - 3 atoms bonded by central atoms, 0 Lone Pair. Refer to the Figure 2.7
Type of molecule AB3
Formula example BF3
Lewis Structure
Polarity: Nonpolar
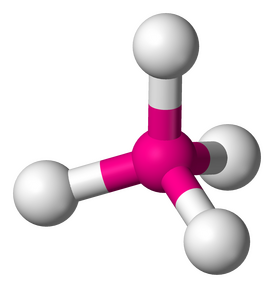
3. TETRAHEDRAL - 4 atoms bonded by central atoms, 0 Lone Pair. Refer to the Figure 2.8
Type of molecule: AB4
Formula example: CH4
Lewis Structure
Polarity: Nonpolar
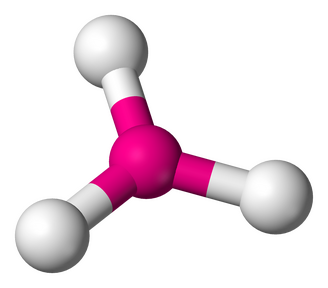
4. TRIGONAL PYRAMIDAL - 3 atoms bonded by central atoms, 1 Lone Pair. Refer to the Figure 2.9
Type of molecule: AB3E Formula example: NH3 lewis Structure Polarity: Polar
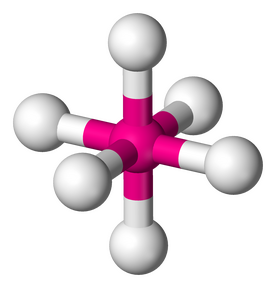
5. TRIGONAL BIPYRAMIDAL - 5 atoms bonded by central atoms 0 Lone Pair. Refer to Figure 2.10
Type of moleculeAB5
Formula example PCl5
Lewis Structure
Polarity: Nonpolar
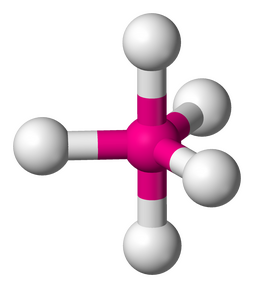
6. OCTAHEDRAL - 6 atoms bonded by central atoms, 0 Lone Pair. Refer to Figure 2.11
Type of molecule AB6
Formula example SF6
Lewis Structure
Polarity: Nonpolar
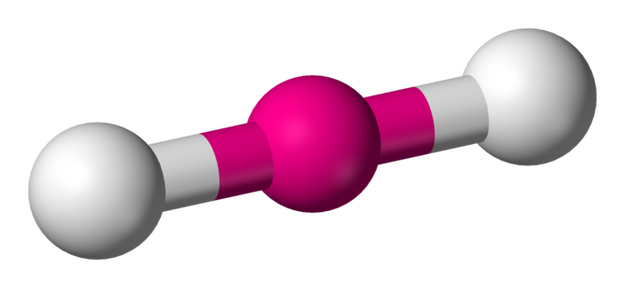
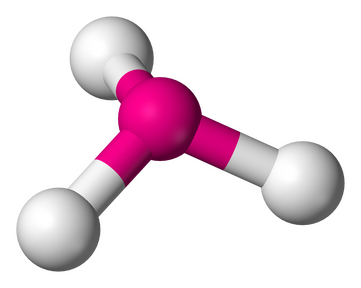
7. BENT OR ANGULAR - 2 atoms bonded by central atoms 2 Lone Pair
Type of molecule AB2E2. Refer to Figure 2.12
Formula example H2O
Lewis Structure
Polarity: Polar